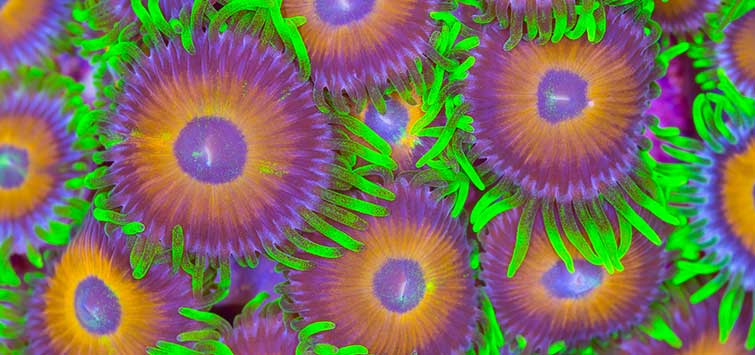
Aquarium Science: Palytoxin and You: How and Why to Avoid a Deadly Zoanthid Toxin
Author: Nicholas Violand
If you've never heard of palytoxin, you need to know it's a seriously dangerous substance found in some ornamental reef polyps—read about how this toxin kills, and why you’re always better safe than sorry.
Dangerous Palytoxin
Marine environments harbor organisms containing novel chemical compounds, such as green fluorescent protein or different types of luciferins from bioluminescent organisms like jellyfish. Such compounds can play a unique and important role in scientific research, and these two are also what can make an organism glow. On the other hand, marine organisms may contain toxins which, while interesting to study, can be deadly. Palytoxin, from a Palythoa zoanthid, was isolated in 1971 by scientists in Hawai‘i.
Although Moore and Scheuer, the scientists who discovered it, reported the LD50 to be roughly 15 nanograms per kilogram in mice (which, in toxicological terms, is a dosage concentration—in this case, a very minute one—that kills 50 percent of a tested population in a given time), very little was originally known about the compound or the organism in which it was found. Nor was the species described in 1971, but it eventually came to be known as P. toxica, and is currently said to carry one of the most toxic natural substances known to man—and coincidentally, zoanthids are some of the most common marine invertebrates kept by reef aquarists.
With more information, it’s hopeful that reefkeepers will exercise caution when handling any marine invertebrates—for although poisonings may be uncommon, they’re not impossible.
The Legend of Palytoxin
This organism was found by native Hawai‘ians and used for its unique toxic properties some years prior to the discovery of palytoxin, and a bit of mythology surrounded it. The legend said that before warriors collected limu make ‘o Hana, falsely called “the deadly seaweed of Hana,” from that same tidepool to create lethal poison-tipped spears, it was believed that a mysterious murderer inhabited the area. When fishermen began to disappear, some of the locals began to suspect a lone, hump-backed farmer.
When he was confronted, it was discovered that the hump on his back was actually the mouth of a shark fused to the man—and this was a shark god. The culprit was expediently killed and his body burned; his ashes were spread across the tidepool, and in that location, everything had become cursed, or otherwise toxic. What was called seaweed by those who collected it for its toxin is actually a brown palytoxin-producing zoanthid, which is said to still fill the tidepool. Scientists have since been researching the complex organic compound found in zoanthid polyps in attempts to better understand how it works.
Findings in Other Species
Since palytoxin was discovered in Hawai‘i, it has also been found in other species, such as P. tuberculosa near Ishigaki Island in the Ryukyus of Japan, and P. caribaeorum and P. mammilosa in Jamaica and the Bahamas (Moore and Scheuer, 1971). It was also isolated in P. vestitus from Hawai‘i, and other unidentified species in Tahiti—even sea anemones, crabs, and a dinoflagellate (one-celled organism) (Oku, 2004). The toxin is no longer as isolated as originally thought, and to make matters worse, most zoanthid specimens can be difficult to identify on the species or even genus levels. Thanks to advancements in science and technology, and the overall ingenuity of the hobby, marine aficionados from all over the globe that had once warily kept marine invertebrates are now mastering the husbandry of many coral-reef organisms—among them various zoanthids of the genera Zoanthus and Palythoa, particularly recommended to beginning reefkeepers for their ease of care.
Caution Regarding Exotic Creatures
While the particular species that house palytoxin may not be widely available in the trade, there are around 60 species (likely more are undescribed) of Palythoa, Protopalythoa, Zoanthus, and Parazoanthus, so the difficulty in identifying toxic species makes this impossible to verify. However, I’ve heard from one expert who says they are more common than I originally thought—so common that he was able to send me some toxic zoanthids from his stock, believed to actually be P. toxica. So caution is always recommended when coming into contact with any such exotic creatures.
Properties of Palytoxin
Palytoxin is a rather large molecule which, since its discovery, has been artificially synthesized by two separate groups of scientists (Kishi, 1989). When isolated, it is a white, amorphous solid that is soluble in water, which leads to its quick action in the body. If administered at the right dose in mice, it can kill in minutes. The compound was also noted as having a half-life of 55 to 85 minutes, indicating that it may degrade under certain circumstances such as the acidic environment of the human stomach. Keep in mind, although this half-life seems short, once in the body, palytoxin doesn’t need much time to take effect (Moore and Scheuer, 1971).
Toxicokinetics
The complex structure of this compound, having a molecular formula of C129H223N3O54, can take effect in any way that it is administered, as it attacks the Na/K (sodium/potassium) pumps of most animal cells—more on this later. It can be taken into the body through openings or cuts in the skin, by ingestion, or injection, as was done in the original study of the toxin.
The post-mortem analysis of an East Timorese man who died hours after eating a crab Zosimus aeneus, a species also known to harbor palytoxin, among others, had been conducted a few years ago (Llewellyn et al., 2002.) While palytoxin was not found in any of the body’s fluids, the man died from ingesting another marine toxin, known as a paralytic shellfish toxin, only hours after ingestion.
So aside from purposely ingesting the mucus excreted from a Palythoa sp. or the polyps themselves, anyone handling zoanthids runs the risk of unknowingly picking up this or possibly other toxins on his or her hands, especially after touching an agitated or recently fragged specimen. As previously mentioned, it can enter through cuts in the skin very easily, and can also manifest as a rash on the skin if not taken up into the body.
In the Body
Once in the body, the compound may travel through the bloodstream, and can attack cells at will. A team of scientists tested the compound with rat cells, after noting that bacterial and fungal cells did not seem to be affected. After applying concentrated palytoxin, they observed the cells swelling into globular shapes, along with gradual shrinkage of the nuclei, and the cells burst shortly thereafter (Oku et al., 2004). This same action is noted in various mammalian blood cells, in which disintegrative activity has been observed. The toxin does not need to enter the cell to take its effect, as it affects cells from the outside via the Na/K pump, letting an endless flow of sodium and potassium ions into the cell.
Toxicodynamics
While Moore and Scheuer’s original LD50 of 15 nanograms per kilogram in mice has been debated, it has been speculated that a dose of 100 nanograms per kilogram is fatal in humans. So theoretically, a barely visible amount of isolated palytoxin (about nine thousandths of a percent of an aspirin tablet) is enough to kill an average man. It was also found to be most potent by intravenous injection, and three times less effective by subcutaneous exposure, as through a cut in the skin.
Somewhat fortunately, the least toxic administration method was intragastric, although human fatalities by ingestion have been known; this is typically due to the process of biomagnification, by which other organisms that live in close association with—or consume—the zoanthid colonies pick up the toxin, magnifying it in concentration.
For example, the crab species mentioned earlier may carry palytoxin, and human ingestion of the crab can lead to fatality. Had the man ingested palytoxin and not paralytic shellfish toxin, there is a good chance he would have died within minutes, rather than hours. I have also read about aquarists’ experiences with contact dermatitis reactions from zoanthids, but these are questionably associated with palytoxin—though dermatitis is definitely another reason to be careful with zoanthids, a reason, I’d say, to wear rubber gloves when handling them.
Symptoms
Symptoms of palytoxin exposure are vasoconstriction (narrowing of blood vessels), hemorrhage, ataxia (inability to coordinate voluntary muscle contraction), muscle weakness, ventricular fibrillation (uncoordinated contraction of the cardiac muscle of the ventricles in the heart), ischemia (restriction in blood supply), and eventually death. This restricted blood supply can also be tied to an overall decrease in blood pressure. These symptoms all point to a cardiovascular assault, and regardless of how the toxin is administered, it seems to manifest as a restricted blood supply to the rest of the body, leading to death.
Mechanism of Action
To finally explain this sodium and potassium business, the Na/K pumps help to establish normal sodium and potassium gradients across outer membranes of most animal cells. They also happen to be specific targets for two natural toxins: heart glycosides and palytoxin. Without this gradient, heart cells cannot function properly; but the heart glycosides, such as those produced by the foxglove plant Digitalis purpurea, differ greatly from palytoxin. They also function differently, and believe it or not, Digitalis is used at low doses to treat heart problems.
Palytoxin changes the Na/K pump into a non-specific ion channel, completely destroying the ion gradient that cells rely upon for transport and maintenance of overall cell volume. This is inevitably what leads to cell death in heart tissue, which kills the victim so quickly. The destruction of heart tissue leads to a loss of blood and oxygen to the rest of the body, and death essentially occurs from suffocation.
Treatment?
While it has been suggested that a vasodilator may be an effective treatment if immediately injected into the ventricle of the heart, the reliability of this treatment is not good, especially as poisoning with this toxin may not be known until post-mortem analysis. Typically, an individual afflicted with severe palytoxin poisoning would not be recognized as such in a significant amount of time to administer any sort of effective treatment. It must also be kept in mind that the previously mentioned studies involve a chemically extracted, purified form of palytoxin, and what is present in the mucus of such zoanthids is not as concentrated, yet still dangerous.
Of course, the best treatment for any condition is prevention. Reefkeepers handling any marine invertebrates should use great caution, and gloves are highly recommended for zoanthids and corals, whether the specimen is known to be dangerous or not. Tools, like a pair of tongs, are even better for moving and handling specimens. And while palytoxin may not be present in all zoanthids, various reports of negative reactions abound, especially since any allergic response will vary from person to person. Even worse, palytoxin is thought to be carcinogenic in humans, acting as a skin tumor promoter (Wattenberg, 2006)—another reason to wear gloves!
Take Proper Precautions
Interestingly enough, the elucidation of the origin of palytoxin has not yet arisen. Since its discovery, we’ve blindly assumed it is produced by the zoanthids themselves, but there has been speculation that zoanthids are simply in the chain of biomagnification, and that the toxin is made by a dinoflagellate known as Ostreopsis siamensis. Collecting the toxin, the zoanthids may be concentrating it for other uses like defense. This does not seem at all surprising, considering the symbiotic relationship between many corals and the photosynthesizing zooxanthellae they depend upon. Additionally, while this toxin was assumed to be extremely rare, occurring in merely one location in the world, it has since been found in numerous locations
Thankfully, for any reefkeeper that could potentially house dangerous species of Palythoa, accidental ingestion seems to be the least toxic form of administration of the toxin, and entry through the skin is not nearly as toxic as entry directly into the bloodstream, which is the least likely form of intoxication. However, due to the discovery of palytoxin in other organisms, we know it can occur in organisms such as crabs through biomagnification. If such organisms are ingested, the risk of death is high, but more likely due to the accumulation of other, more-common marine toxins.
So in terms of palytoxin poisoning, we should all understand that caution is necessary around zoanthids, regardless of the presence or absence of this toxin. Don’t handle them with open cuts, and always wash your hands well after touching them. Wearing gloves is an even smarter way to prevent toxic troubles, and goggles or protective eyewear for when cutting frags. And if you think you’ve experienced some sort of allergic reaction, consult with your physician. Although most reefkeepers aren’t fostering palytoxin production, unless he or she is a taxonomist or marine biologist, it may be nearly impossible to know for sure.
Acknowledgments
Special thanks to Leroy and Sally Jo Headlee at the Geothermal Aquaculture Research Foundation for their assistance and expertise.
See the full article on TFH Digital http://www.tfhdigital.com/tfh/200805/#pg122

.png?h=595&iar=0&w=2781&hash=5FD5E69473BCC22199FBFA2FB71B6033)